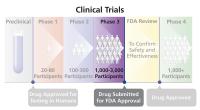
Phase 3 Trial
Audio
The third step in testing an experimental drug (or other treatment) in humans. Phase 3 trials are conducted to confirm and expand on safety and effectiveness results from Phase 1 and 2 trials, to compare the drug to standard therapies for the disease or condition being studied, and to evaluate the overall risks and benefits of the drug. This trial phase recruits a large group of people with the disease or condition, usually ranging from 1,000 to 3,000 participants. The U.S. Food and Drug Administration (FDA) reviews results from Phase 3 trials when considering a drug for approval.