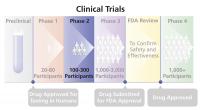
Phase 2 Trial
The second step in testing an experimental drug (or other treatment) in humans. Typically, Phase 2 trials are done only if Phase 1 trials have shown that the drug is safe, but sometimes Phase 1 and Phase 2 trials are combined. Phase 2 trials are designed to evaluate the drug's effectiveness in people with the disease or condition being studied and to determine the common short-term adverse effects and risks associated with the drug. Phase 2 trials involve more participants (often several hundred) and typically last longer than Phase 1 trials.