Drug information
| vesatolimod.mp3 |
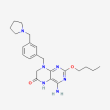
C22 H30 N6 O2
4-amino-2-butoxy-8-[[3-(pyrrolidin-1-ylmethyl)phenyl]methyl]-5,7-dihydropteridin-6-one
Vesatolimod is in Phase 2a development for HIV treatment.
vesatolimod
Molecular Weight: 410.5
(Compound details obtained from PubChem,1 Gilead Sciences website,2 Clinical Infectious Diseases article,3 and ClinicalTrials.gov4,5)
Pharmacology
Mechanism of Action
Immune modulator. Vesatolimod is a selective toll-like receptor 7 (TLR7) agonist. It is an orally administered small molecule that works by binding to and activating TLR7, a pattern-recognition receptor (PRR) that is expressed primarily on plasmacytoid dendritic cells (pDCs) and B cells. PRRs are a key component of the innate host defense mechanism as they recognize and respond to infectious pathogens. As an immune modulator, vesatolimod may contribute to HIV control by triggering numerous inflammatory and immune responses, such as induction of interferon-stimulated gene expression and cytokine production, including type I interferon-alfa (IFN-alfa), as well as activation of natural killer (NK) cells, phagocytic cells, pDCs, and T cells.3,6–9
Vesatolimod’s potential role as a latency-reversing agent has also been evaluated in preclinical and clinical trials, with mixed results.3,6,7,10,11 Mechanistically, research has shown that the induction of HIV RNA by vesatolimod is dependent on IFN-alfa production by pDCs.6
Half-life (T½)
In a Phase 1b dose-escalation trial (NCT02858401) of vesatolimod (1 to 12 mg) administered orally every two weeks in adults with HIV who were virologically suppressed on ART, the vesatolimod median half-life ranged from 9 to 19 hours.3
Metabolism/Elimination
Vesatolimod undergoes metabolism by CYP3A enzymes.12
Select Clinical Trials
Study Identifiers: A5374; NCT06071767
Sponsor: National Institute of Allergy and Infectious Diseases (NIAID)
Phase: 1/2a
Status: This study is currently recruiting participants.
Study Purpose: The purpose of this study is to evaluate the safety and efficacy of a combination regimen in adults who initiated suppressive ART during acute HIV. The combination regimen contains (1) ChAdV- and MVA-vectored conserved mosaic T-cell vaccines, (2) vesatolimod, and (3) broadly neutralizing antibodies 3BNC117-LS (also known as teropavimab or GS-5423) and 10-1074-LS (also known as zinlirvimab or GS-2872).
Study Population:
- Participants are adults with HIV who initiated ART within 90 days of acute HIV diagnosis and who have not interrupted ART for more than 14 consecutive days since initiation of ART.
- Participants have been receiving ART with an INSTI-based regimen with two NRTIs or a dolutegravir/lamivudine regimen for at least 6 weeks prior to study entry.
- Participants have HIV RNA <50 copies/mL since initial viral suppression on ART and for at least 1 year and within 60 days prior to study entry.
- Participants have CD4 counts ≥500 cells/mm3 within 60 days of study entry.13
Study Identifiers: AELIX-003; NCT04364035
Sponsor: Aelix Therapeutics
Phase: 2a
Status: This study has been completed.
Study Purpose: The purpose of this study was to evaluate the safety, immunogenicity, and efficacy of therapeutic HIV vaccines (HIVACAT T-cell immunogen [HTI] vaccines MVA.HTI and ChAdOx1.HTI) and vesatolimod in adults who had received early ART treatment.
Study Population:
- Participants were adults with HIV who had initiated ART within 6 months of HIV acquisition.
- Participants had HIV RNA <50 copies/mL for at least 1 year before screening and at screening.
- Participants had CD4 counts ≥450 cells/mm3 for at least 6 months prior to screening and had nadir CD4 counts ≥200 cells/mm3 since HIV diagnosis.4
Selected Study Results: Results presented at CROI 2023 and IAS 2023 and published in Nature Communications (2025) showed that the combination of HTI vaccines with 10 doses of vesatolimod was safe with only one serious adverse event (SAE) reported, which was unrelated to the study drugs. HTI vaccines given with vesatolimod proved to be highly immunogenic. Vaccine-induced HTI-specific T-cell responses contributed to improved viral control after participants underwent an analytical treatment interruption (ATI) of ART.14-16
Additional Published Material:
- Commun Med (Lond) article, 2025: Predictors of virological outcomes after analytical interruption of antiretroviral therapy and HTI vaccination in early treated people with HIV-1
Study Identifiers: GS-US-382-5445; NCT05281510
Sponsor: Gilead Sciences
Phase: 2a
Status: This study has been completed.
Study Purpose: The purpose of this open-label trial was to evaluate the safety and tolerability of the bNAbs VRC07-523LS and CAP256V2LS in combination with vesatolimod in early ART-treated women with clade C HIV.
Study Population:
- Participants were adult females with HIV who were recruited from the Females Rising through Education, Support, and Health (FRESH) acute HIV infection cohort.
- Participants had been receiving ART for at least 12 consecutive months prior to screening.
- Participants had HIV RNA <50 copies/mL for 12 consecutive months prior to screening and at screening.
- Participants had CD4 counts ≥500 cells/mm3.
- Participants had documented viral sensitivity to VRC07-523LS or CAP256V2LS.5
Selected Study Results: Results presented at CROI 2025 and EACS 2025 showed that vesatolimod and the bNAbs VRC07-523LS and CAP256V2LS were safe, with no treatment-related SAEs reported. Eighteen participants (90%) experienced infusion-related reactions, most of which were mild in severity and resolved within 2 days. One participant had a treatment-emergent AE resulting in discontinuation of vesatolimod. During an ATI of ART, varying patterns of viral control were seen among the 20 participants.17,18
Additional Published Material:
- IAS, 2025: Vesatolimod pharmacodynamic responses in a trial of vesatolimod and broadly neutralizing antibodies in early-treated South African women with clade C HIV-1
Additional early-phase trials evaluating vesatolimod for HIV treatment have been conducted, including:
- GS-US-382-1450 (NCT02858401): A Phase 1b dose-escalation study that evaluated the safety and virologic effect of vesatolimod in adults with HIV who had viral suppression on ART. This study has been completed, and results are published in Clin Infect Dis (2021).19
- GS-US-382-3961 (NCT03060447): A Phase 1b study that evaluated the safety and efficacy of vesatolimod in adults with HIV who had undetectable or low viral load levels (between 50 and 5,000 copies/mL) before initiating ART and viral suppression on ART. This study has been completed, and results are available from Sci Transl Med (2021) and CROI 2023.20
Adverse Events
AELIX-003 (NCT04364035)
In this Phase 2a trial, 33 participants received HTI vaccines plus vesatolimod and 17 participants received placebo. Investigators reported that approximately ninety-one percent of participants receiving vaccines plus vesatolimod and 65% of participants receiving placebo experienced a treatment-emergent AE related to study drugs. All treatment-emergent AEs related to study drugs were Grade 1 or 2 in severity. Among the participants receiving HTI vaccines plus vesatolimod, the most common treatment-emergent AEs related to study drugs were injection-site pain (87.9%), influenza-like illness (36.4%), headache (33.3%), and fatigue (33.3%). None of the drug-related AEs led to treatment discontinuation. No SAEs related to the study drugs occurred, and no cases of cytokine-release syndrome were reported.4,14,16
GS-US-382-5445 (NCT05281510)
In this Phase 2a trial, 20 participants received up to 10 oral doses of vesatolimod and IV infusions of the bNAbs VRC07-523LS and CAP256VLS. Treatment-related AEs, most of which were mild, occurred in 18 participants (90%). Two participants had Grade 3 treatment-related AEs (fever and chills). The most common treatment-related AEs were infusion-related reactions (90%), headache (65%), tachycardia (40%), fever (35%), and chest discomfort (25%). The majority of treatment-related infusion reactions associated with the bNAbs were mild and all resolved within 2 days. Most of the treatment-related AEs associated with vesatolimod were mild or moderate and occurred after the first dose. No treatment-related SAEs were reported. One participant developed a treatment-related AE—Grade 1 cytokine release syndrome—that led to discontinuation of vesatolimod.17,18
GS-US-382-1450 (NCT02858401)
In this Phase 1b dose-escalation trial, 48 participants received multiple doses of either vesatolimod (1 to 12 mg) (n = 36) or placebo (n = 12) administered once every other week. At least one adverse event (AE) occurred in 64% of participants who received vesatolimod and 75% of participants who received placebo. AEs related to vesatolimod treatment included fatigue, headache, myalgia, and pyrexia. Drug-related AEs occurred more frequently in the 8-mg and 10/12-mg vesatolimod dose groups compared to the placebo group. Study drug-related AEs were mild, resolved within 1–2 days, and did not occur with each dose. There were no drug-related SAEs or discontinuations due to an AE. The majority of laboratory abnormalities that occurred during the study were Grade 1 or 2.3
GS-US-382-3961 (NCT03060447)
In this Phase 1b trial, 25 participants were randomized to receive 10 doses of vesatolimod (4 to 8 mg) (n = 17) or placebo (n = 8) administered once every other week. Ninety-four percent of participants receiving vesatolimod and 75% of participants receiving placebo had at least one AE, most of which were mild to moderate in severity. Drug-related AEs occurred in 53% of participants in the vesatolimod group and 13% of participants in the placebo group. The majority of AEs related to vesatolimod were mild, transient, influenza-like symptoms — chills, headache, lymphadenopathy, fatigue, and pyrexia. One participant receiving vesatolimod experienced a Grade 2 treatment-emergent SAE — chronic gastritis. Another participant receiving vesatolimod had Grade 3 drug-related cases of gout, arthralgia, and sciatica, all of which resolved without treatment. There were no discontinuations due to drug-related AEs. Laboratory abnormalities were mostly Grade 1 or 2 in severity.7
Drug Interactions
Vesatolimod is metabolized via the CYP3A pathway and is a substrate of the drug transporters P-gp and BCRP. In a Phase 1 drug-drug interaction study (NCT05458102), investigators evaluated the effects of vesatolimod (subtherapeutic doses) coadministered with cobicistat (a P-gp, BCRP, and strong CYP3A inhibitor) or voriconazole (a strong CYP3A inhibitor) on vesatolimod pharmacokinetics/pharmacodynamics in adults with HIV on ART. Results indicated that vesatolimod coadministered with vorinostat did not alter vesatolimod pharmacokinetics. However, vesatolimod coadministered with cobicistat increased vesatolimod plasma concentrations.12
References
- National Center for Biotechnology Information. PubChem compound summary for CID 46241268, vesatolimod. Accessed February 6, 2026
- Gilead Sciences website. Pipeline. Accessed February 6, 2026
- Riddler SA, Para M, Benson CA, et al. Vesatolimod, a toll-like receptor 7 agonist, induces immune activation in virally suppressed adults living with human immunodeficiency virus–1. Clin Infect Dis. 2021;72(11):e815-e824. doi:10.1093/cid/ciaa1534. Accessed February 6, 2026
- Aelix Therapeutics. A Phase IIa randomised, double-blind, placebo-controlled study of HIV-1 vaccines MVA.HTI and ChAdOx1.HTI with TLR7 agonist vesatolimod (GS-9620) in early treated HIV-1 infection. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on March 5, 2020. NLM Identifier: NCT04364035. Accessed February 6, 2026
- Gilead Sciences. A Phase 2a study to evaluate the safety and tolerability of a regimen of dual anti-HIV envelope antibodies, VRC07-523LS and CAP256V2LS, in a sequential regimen with a TLR7 agonist, vesatolimod, in early antiretroviral-treated HIV-1 clade C-infected women. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on March 7, 2022. NLM Identifier: NCT05281510. Accessed February 6, 2026
- Tsai A, Irrinki A, Kaur J, et al. Toll-like receptor 7 agonist GS-9620 induces HIV expression and HIV-specific immunity in cells from HIV-infected individuals on suppressive antiretroviral therapy. J Virol. 2017;91(8):e02166-16. doi:10.1128/JVI.02166-16. Accessed February 6, 2026
- SenGupta D, Brinson C, DeJesus E, et al. The TLR7 agonist vesatolimod induced a modest delay in viral rebound in HIV controllers after cessation of antiretroviral therapy. Sci Transl Med. 2021;13(599):eabg3071. doi:10.1126/scitranslmed.abg3071. Accessed February 6, 2026
- Bam RA, Hansen D, Irrinki A, et al. TLR7 agonist GS-9620 is a potent inhibitor of acute HIV-1 infection in human peripheral blood mononuclear cells. Antimicrob Agents Chemother. 2016;61(1):e01369-16. doi:10.1128/AAC.01369-16. Accessed February 6, 2026
- Rebbapragada I, Birkus G, Perry J, Xing W, Kwon H, Pflanz S. Molecular determinants of GS-9620-dependent TLR7 activation. PLoS ONE. 2016;11(1):e0146835. doi:10.1371/journal.pone.0146835. Accessed February 6, 2026
- Lim SY, Osuna CE, Hraber PT, et al. TLR7 agonists induce transient viremia and reduce the viral reservoir in SIV-infected rhesus macaques on antiretroviral therapy. Sci Transl Med. 2018;10(439):eaao4521. doi:10.1126/scitranslmed.aao4521. Accessed February 6, 2026
- Del Prete GQ, Alvord WG, Li Y, et al. TLR7 agonist administration to SIV-infected macaques receiving early initiated cART does not induce plasma viremia. JCI Insight. 4(11):e127717. doi:10.1172/jci.insight.127717. Accessed February 6, 2026
- Zheng Y, Wire M, Omange RW, et al. The effects of cobicistat and voriconazole on the safety, pharmacokinetics, and pharmacodynamics of the TLR7 agonist vesatolimod in people with HIV. Infect Dis Ther. 2025;14(11):2535-2549. doi:10.1007/s40121-025-01227-x. Accessed February 6, 2026
- National Institute of Allergy and Infectious Diseases (NIAID). A Phase I/IIa randomized, placebo-controlled trial of conserved-mosaic T-cell vaccine in a regimen with vesatolimod and broadly neutralizing antibodies in adults initiated on suppressive antiretroviral therapy during acute HIV-1. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on September 5, 2023. NLM Identifier: NCT06071767. Accessed February 6, 2026
- Mothe B, Curran A, de Quirós JCLB, et al. A placebo-controlled randomized trial of the HTI immunogen vaccine and vesatolimod. Poster presented at: Conference on Retroviruses and Opportunistic Infections (CROI); February 19-22, 2023; Seattle, WA. Poster 433. Accessed February 6, 2026
- Mothe B, Bailon L, Curran A, et al. T-cell responses induced by HTI vaccines and vesatolimod correlate with improved control of HIV rebound. Abstract presented at: International AIDS Society (IAS) Conference on HIV Science; July 23-26, 2023; Brisbane, Australia. Abstract LBEPB20. Accessed February 6, 2026
- Bailón L, Moltó J, Curran A, et al. Safety, immunogenicity and effect on viral rebound of HTI vaccines combined with a TLR7 agonist in early-treated HIV-1 infection: a randomized, placebo-controlled phase 2a trial. Nat Commun. 2025;16:2146. doi:10.1038/s41467-025-57284-w. Accessed February 6, 2026
- Dong K, Asari V, Govender V, et al. Evaluation of 2 bNAbs plus vesatolimod in early-treated South African women with HIV-1 during ATI. Abstract presented at: Conference on Retroviruses and Opportunistic Infections (CROI); March 9-12, 2025; San Francisco, CA. Abstract 105. Accessed February 6, 2026
- Lim D, Fu Olivia, Liu Xiaopeng, et al. Safety and analytical treatment interruption outcomes in a clinical trial of 2 broadly neutralizing antibodies plus vesatolimod in early-treated South African women with clade C HIV-1. European AIDS Conference; October 15-18, 2025; Paris, France. Conference reports for National AIDS Treatment Advocacy Project (NATAP); 2025. Accessed February 6, 2026
- Gilead Sciences. A Phase 1b, randomized, blinded, placebo-controlled dose-escalation study of the safety and biological activity of GS-9620 in HIV-1 infected, virologically suppressed adults. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on July 26, 2016. NLM Identifier: NCT02858401. Accessed February 6, 2026
- Gilead Sciences. A Phase 1b, randomized, double-blind, placebo-controlled study to evaluate the safety and efficacy of GS-9620 in antiretroviral treated HIV-1 infected controllers. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on February 17, 2017. NLM Identifier: NCT03060447. Accessed February 6, 2026
Last Reviewed: February 6, 2026