Drug information
| MK-8527.mp3 |
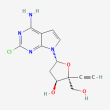
C13 H13 Cl N4 O3
(2,3,5)-5-(4-amino-2-chloropyrrol[2.3-d]pyrimidin-7-yl)-2-ethinyl-2-(hydroxymethyl) oxolan-3-ol
MK-8527 is in phase III development for HIV prevention. (MK-8527 has also been previously studied for HIV treatment.)
MK-8527
Molecular Weight: 308.72
(Compound details obtained from PubChem,1 Merck website,2 ClinicalTrials.gov,3,4 and CROI 2024 abstract5)
Pharmacology
Mechanism of Action
Nucleoside reverse transcriptase translocation inhibitor (NRTTI). MK-8527, a deoxyadenosine analog, is a unique compound derived through structural modification of the benchmark NRTTI islatravir. In vitro, MK-8527 has shown potent and specific activity against HIV-1 and has also exhibited activity against a panel of HIV-1 subtypes and HIV-2.6,7
Intracellularly, MK-8527 is converted to its active triphosphate form (MK-8527-TP). As an NRTTI, MK-8527-TP inhibits HIV reverse transcriptase (RT) through multiple modes of action. Once incorporated into viral DNA, MK-8527-TP functions as an immediate chain terminator by blocking RT translocation. In instances where RT translocation does occur and an additional nucleotide is allowed to incorporate onto the viral DNA chain, MK-8527-TP acts as a delayed chain terminator by causing structural changes to the viral chain.6,7
MK-8527 is currently in Phase 3 development as a once-monthly oral tablet for HIV pre-exposure prophylaxis (PrEP).3,4
Half-life (T½)
In a preclinical study of orally administered MK-8527 (50 mg/kg) in rhesus monkeys, the plasma half-life of the parent compound was approximately 7 hours and the intracellular half-life of the active triphosphate (MK-8527-TP) in peripheral blood mononuclear cells (PBMCs) was approximately 48 hours.7
In a Phase 1 study (MK‐8527‐003) of multiple ascending weekly oral doses of MK-8527 (5–40 mg) in healthy adult participants, the apparent terminal half-life of MK-8527-TP was 216–291 hours across all dose levels.8
Select Clinical Trials
Study Identifiers: MK-8527-007; NCT06045507
Sponsor: Merck Sharp & Dohme LLC
Phase: 2a
Status: This study has been completed.
Study Purpose: The purpose of this study was to evaluate the safety, tolerability, and pharmacokinetics of once-monthly oral MK-8527 given at 3 different dose levels in participants at low risk for HIV.
Study Population:
- Participants were adults without HIV who had a low likelihood of acquiring HIV.
- Participants had no prior use of MK-8527 or islatravir.9
Selected Study Results: Results presented at IAS 2025 demonstrated that MK-8527, at all doses studied, was generally well tolerated and had a safety profile comparable to placebo. The pharmacokinetics of MK-8527 in plasma and MK-8527-TP in PBMCs were dose proportional.10
Study Identifiers: EXPrESSIVE-10; MK-8527-010; NCT07071623
Sponsor: Merck Sharp & Dohme LLC
Phase: 3
Status: See the ClinicalTrials.gov record for this study’s status.
Study Purpose: The purpose of this study is to evaluate the safety and efficacy of once-monthly oral MK-8527 as HIV PrEP in sexually active women and adolescent girls in sub-Saharan Africa.
Study Population:
- Participants are sexually active women and adolescent girls (16 to 30 years of age) without HIV.
- Participants have never taken cabotegravir, lenacapavir, or any other long-acting HIV PrEP product in the past. 4,11
Study Identifiers: EXPrESSIVE-11; MK-8527-011; NCT07044297
Sponsor: Merck Sharp & Dohme LLC
Phase: 3
Status: This study is currently recruiting participants.
Study Purpose: The purpose of this study is to evaluate the safety and efficacy of once-monthly oral MK-8527 as HIV PrEP in sexually active adults and adolescents who could benefit from PrEP.
Study Population:
- Participants are people without HIV who have a higher likelihood of HIV exposure.
- Participants have never taken cabotegravir, lenacapavir, or any other long-acting HIV PrEP product in the past.
- See the ClinicalTrials.gov record for additional information on eligibility criteria.3,11
Other studies of MK-8527 are ongoing or planned, including the following Phase 1 trials:
- MK-8527-009 (NCT06580587): An open-label study evaluating drug concentrations of MK-8527 in breast milk following a single oral dose in healthy lactating female participants. This study is currently recruiting participants.12
- MK-8527-014 (NCT07063238): A study evaluating the effect of a single oral supratherapeutic dose of MK-8527 on the QTc interval in healthy adults. This study is currently recruiting participants.13
- MK-8527-015 (NCT07025551): An open-label trial evaluating the pharmacokinetics of a single oral dose of MK-8527 in participants with mild or moderate hepatic impairment. This study is currently recruiting participants.14
Two additional Phase 1 trials (MK-8527-001 and MK-8527-003) evaluating the safety and pharmacokinetics of ascending single and multiple oral doses of MK-8527 in adults without HIV have been completed. Results are available from Clin Transl Sci (2025).8
Adverse Events
MK-8527-007 (NCT06045507)
In this Phase 2a study, adults without HIV received oral monthly doses of MK-8527 (3 mg [n = 101], 6 mg [n = 101], or 12 mg [n = 99]) or placebo (n = 49) for up to 6 months. The safety profile of MK-8527 at all doses studied was similar to that of placebo. The majority of drug-related adverse events (AEs) were Grade 1 or 2 and resolved during the study. One participant in the MK-8527 3-mg dose group experienced a drug-related serious adverse event (SAE) of spontaneous abortion. Two participants discontinued MK-8527 because of a drug-related AE—one participant receiving MK-8527 6 mg due to Grade 1 decreases in CD4/lymphocyte counts and another participant receiving MK-8527 12 mg due to Grade 2 hypoesthesia.9,10
Participants who met criteria for study drug discontinuation due to reductions in CD4 and/or lymphocyte counts, as specified by the study protocol, had their levels return to baseline within 11 weeks. Overall, the changes in CD4 and total lymphocyte counts seen with MK-8527 were deemed clinically insignificant.10
Drug Interactions
A Phase 1 drug-drug interaction trial (NCT06783192) evaluated the effects of oral MK-8527 on the pharmacokinetics (PK) of the oral hormonal contraceptive levonorgestrel (LNG)/ethinyl estradiol (EE) in adult (18 to 70 years of age) female participants without HIV. Results demonstrated that coadministration of MK-8527 with LNG/EE had no significant effect on the PK of LNG or EE.15,16
References
- National Center for Biotechnology Information. PubChem Compound Summary for CID 118389446. Accessed September 10, 2025
- Merck website. Our pipeline at a glance. Accessed September 10, 2025
- Merck Sharp & Dohme LLC. A Phase 3, randomized, active-controlled, double-blind clinical study to evaluate the efficacy and safety of MK-8527 oral once-monthly as HIV-1 preexposure prophylaxis. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on June 26, 2025. NLM Identifier: NCT07044297. Accessed September 10, 2025
- Merck Sharp & Dohme LLC. A Phase 3, randomized, active-controlled, double-blind clinical study to evaluate the efficacy and safety of MK-8527 oral once-monthly as HIV-1 preexposure prophylaxis in women. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on July 8, 2025. NLM Identifier: NCT07071623. Accessed September 10, 2025
- Carstens RP, Kapoor Y, Vargo R, et al. Single dose administration of MK-8527, a novel nRTTI, in adults with HIV-1. Abstract presented at: Conference on Retroviruses and Opportunistic Infections (CROI); March 3-6, 2024; Denver, CO. Abstract 115. Accessed September 10, 2025
- Raheem IT, Girijavallabhan V, Fillgrove KL, et al. MK-8527 is a novel inhibitor of HIV-1 reverse transcriptase translocation with potential for extended-duration dosing. PLOS Biol. 2025;23(8):e3003308. doi:10.1371/journal.pbio.3003308. Accessed September 10, 2025
- Raheem I, Fillgrove K S, O’Donnell G, et al. Discovery of MK-8527: a long-acting HIV-1 nucleoside reverse transcriptase translocation inhibitor. Poster presented at: Conference on Retroviruses and Opportunistic Infections (CROI); March 3-6, 2024; Denver, CO. Poster 638. Accessed September 10, 2025
- Gillespie G, Carstens RP, Zang X, et al. Safety and pharmacokinetics of MK‐8527 in adults without HIV. Clin Transl Sci. 2025;18(9):e70331. doi:10.1111/cts.70331. Accessed September 10, 2025
- Merck Sharp & Dohme LLC. A Phase 2a, double-blind, placebo-controlled study to evaluate the safety, tolerability, and pharmacokinetics of oral MK-8527 once monthly in participants at low-risk for HIV-1 infection. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on September 13, 2023. NLM Identifier: NCT06045507. Accessed September 10, 2025
- Mayer K, Kotze P, Lombaard J, et al. Safety and pharmacokinetics of MK-8527 oral once-monthly: a Phase 2 study in adults at low risk of HIV-1 exposure. IAS Conference on HIV Science; July 13-17, 2025; Kigali, Rwanda. Conference reports for National AIDS Treatment Advocacy Project (NATAP); 2025. Accessed September 10, 2025
- Merck: Press release, dated July 14, 2025. Merck to initiate Phase 3 trials for investigational once-monthly HIV prevention pill. Accessed September 10, 2025
- Merck Sharp & Dohme LLC. A clinical study to evaluate the breast milk, plasma and whole blood pharmacokinetics of MK-8527 in healthy lactating female participants. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on August 28, 2024. NLM Identifier: NCT06580587. Accessed September 10, 2025
- Merck Sharp & Dohme LLC. A double-blind, placebo-controlled and active-controlled trial to evaluate the effect of a supratherapeutic dose of MK-8527 on the QTc interval in healthy adult participants. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on July 3, 2025. NLM Identifier: NCT07063238. Accessed September 10, 2025
- Merck Sharp & Dohme LLC. An open-label, single-dose study to evaluate the pharmacokinetics of MK-8527 in participants with mild and moderate hepatic impairment. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on June 9, 2025. NLM Identifier: NCT07025551. Accessed September 10, 2025
- Merck Sharp & Dohme LLC. A study to assess the effects of single oral doses of MK-8527 on the single-dose pharmacokinetics of an oral contraceptive (levonorgestrel and ethinyl estradiol) in healthy adult postmenopausal or oophorectomized female subjects. In: ClinicalTrials.gov. Bethesda (MD): National Library of Medicine (US). Registered on January 14, 2025. NLM Identifier: NCT06783192. Accessed September 10, 2025
- Carstens RP, Kapoor Y, Garrett GC, et al. Phase 1, open-label study to evaluate the drug interaction between MK-8527, an HIV-1 nucleoside reverse transcriptase translocation inhibitor, and the oral contraceptive levonorgestrel/ethinyl estradiol in healthy adult female participants. Poster presented at: International AIDS Conference; July 22-26, 2024; Munich, Germany. Poster WEPEB106. Accessed September 10, 2025
Last Reviewed: September 10, 2025