Drug information
| Symtuza.mp3 |
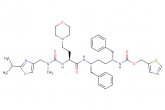
darunavir ethanolate
Molecular Weight: 593.7377
cobicistat
Molecular Weight: 776.0347
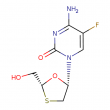
emtricitabine
Molecular Weight: 247.249
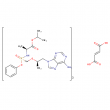
tenofovir alafenamide fumarate
Molecular Weight: 1069.0138
(The drug image[s] shown above is of the brand product only. There may be other available products not shown.)
FDA label informationFDA label information